Immunization Integration Program
Your gateway to immunization interoperability.
-
Increasing interoperability, saving lives
Globally, vaccines save an estimated 2–3 million lives every year. Staying up to date on vaccinations leads to better health outcomes, higher productivity and lower healthcare costs. Improved interoperability and functionality between health tech and immunization information systems (IIS) enhance data quality from the point of care to public health, with the long-term goal of increasing vaccination coverage and reducing vaccine-preventable diseases.
The Immunization Integration Program (IIP) brings together key collaborators, like IIS, EHRs, health information exchanges (HIEs), clinicians, public health, pharmacy and other critical partner groups to improve immunization interoperability. The IIP is a partnership between the Centers for Disease Control (CDC), HIMSS, the American Immunization Registry Association (AIRA) and Drummond Group, and is sponsored by the CDC’s National Center for Immunization and Respiratory Diseases (NCIRD).The pipeline approach
A pipeline approach has been developed to identify opportunities to strategically use the IIP's recommended solutions within the Testing and Recognition initiative. Updates to the test plan are made regularly and include efforts resulting from work by the IIP.
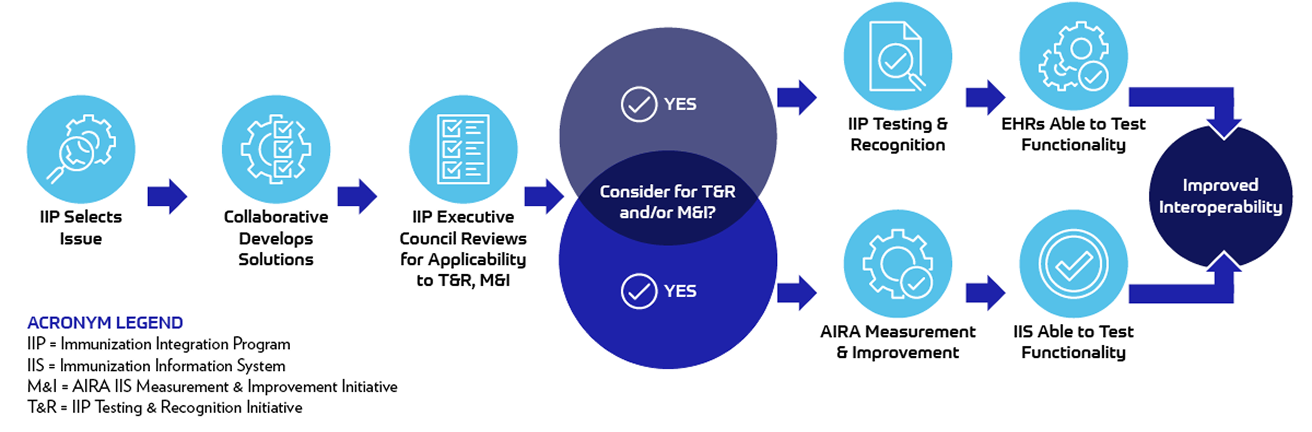
Over time, the test plan will be updated to continue to improve immunization interoperability and information sharing. As immunization data exchange evolves, new testing features will be added and outdated requirements may be removed to ensure the test plan stays current, relevant, and effective.
Collaborate & create solutions
Delivering the right immunization to the right patient at the right time requires a strong immunization program shaped by human interactions, technical capabilities, local workflows, policies and regulations. The IIP offers a platform for participants to share their expertise and challenges while working together to discuss issues, provide perspectives and share solutions. Key participants of the IIP are listed below, and additional groups are always welcome to join.
-
Clinicians
Provide the right vaccines at the right time with better electronic health records (EHR) through IIP's Testing and Recognition program and collaborative activities.
-
Health IT developers
Enhance communication between health IT systems (like EHRs) and immunization information systems (IIS). By sharing best practices, common challenges, and strategies, it will make it easier to connect and share information. As a convener for these two groups, the IIP creates a necessary space for collaboration.
-
HIEs
Increase awareness of how Health Information Exchanges (HIEs) can improve vaccine data sharing and provide valuable member and participant services to a variety of stakeholders, such as healthcare providers, EHRs and IIS.
-
Pharmacies, pharmacy systems and pharmacy associations
Pharmacies play an important role in vaccine delivery — becoming involved in the IIP can support and amplify the pharmacy perspective and foster collaboration with data exchange partners.
-
Public health IIS managers and immunization program managers
Amplify the impact, participation and coverage of immunization programs by tackling issues that will improve CDC goals of improved data quality and EHR-IIS interoperability.
-
IIS technology partners
Collaborate with immunization partners to understand challenges, prioritize enhancements and learn about strategies from the CDC that may impact your development lifecycle.
-
Learn more about the IIP
Learn more about becoming a pilot site, early adopter or workgroup participant and how the IIP is improving immunization interoperability by emailing iip@himss.org.
Partners
-
-
IIP Testing and Recognition Initiative
The IIP Testing & Recognition initiative is a no-cost immunization capabilities testing method enabled by the National Institute of Standards and Technology (NIST) and approved by the Office of the National Coordinator for Health Information Technology (ONC) as an alternative way to meet certain certification criteria. This initiative is one of two strategies to help adopt important immunization features in electronic health records (EHRs), which improves clinical decision-making and information sharing with immunization information systems (IIS). This initiative is an approved testing method for (f)(1) Transmission to immunization registries certification criteria within the Office of the National Coordinator for Health Information Technology (ONC) Health IT Certification Program.
What we’re testing
Eight groups of workflow-related functional requirements will be tested and recognized.
-
Care provision and documentation
Supports end users to prepare, administer and record care provision.
-
Query, response and reconciliation
Supports end users to query, reconcile and view from an immunization registry (IIS).
-
Transmission and acknowledgment
Supports transmission of newly documented patient immunization records to the immunization registry (IIS).
-
Patient access
Allows patients or their caregivers to access immunization records most frequently using a patient portal.
-
Clinical configuration
Supports adding a new vaccine to the EHR.
-
Transport
Supports the ability of the EHR to send and receive immunization messages with an immunization registry (IIS) using the CDC WSDL.
-
Data quality assurance
Supports basic quality checks to evaluate the EHR regarding fundamental data error prevention.
-
Data load
Prepares the EHR for IIP testing.
-

-
Advancing capabilities and functionality
We’re enhancing the exchange of immunization data by developing and implementing advanced functionalities — ensuring more accurate and efficient sharing of immunization information across healthcare systems. Schedule a call to learn more.
-
Enhancing data quality across the board
Help improve the quality of data collected at the point of care and ensure that this high-quality data is accurately and efficiently shared with public health systems by becoming an IIP Testing & Recognition vendor.
-

IIP vendor testimonials
-
The IIP test plan is extremely robust and comprehensive. The test plan includes evidence-based, real world use cases, and will deliver positive results. Each use case facilitates better patient care and ease of use for the provider and in connecting with the IIS.
Chris Gibson
Interoperability Product Director, Office Practicum
-
IIP had the right set of requirements for an EHR who wants to implement excellent immunization workflows. This was important to us and seemed like a good way to get in alignment with best practices and to engage with the community who is making those best practices.
Scott Kirby
Interoperability Developer, PCC - Physicians Computer Company
-
Oracle Health is proud to have participated in this public health initiative to put patients and providers first through improving immunization practices, data sharing, access to historical records and forecasts, and optimal clinical workflows.
Kristen Glaza
Sr. Manager, Product Manager - Oracle Health
Join our leaders
Apply to be an IIP Testing & Recognition vendor and help create improve and inform public health efforts.
-

January 2025: Product tested: Office Practicum OP Version 21 Passed : IIP Test Plan 11.0 Essential Interoperability and Data Quality August 2019: Product tested: Office Practicum OP Version 20 Passed with notable exception: IIP Test Plan 7.6
-

Products tested: • PCC EHR – V9.5.4 Build 22340/Clinical Version V9.5.4 Build 22340 • PCC EHR – V9.7.1 Build 23046/Clinical Version V9.7.1 Build 23054 Passed with notable exception: IIP Test Plan v10.0 (March 2023)
-

Products tested: Cerner FirstNet (Immunizations) Version 2 and PowerChart Immunizations Version 10 Passed with notable exception: IIP Test Plan 5.6 (December 2017)
-

Product tested: • Qvera Interface Engine Version 3.0.44 Build 8640 Passed with notable exception: IIP Test Plan 5.6 (December 2017)
-

Product tested: • GlaceEMR Version 6.0 Passed without exception: IIP Test Plan 5.5 (September 2017)
-
-
IIP Collaborative
The IIP Collaborative fosters an interconnected network of immunization data exchange partners to prioritize, develop and drive the adoption of solutions to improve immunization interoperability. Learn how this group is driving action in the immunization ecosystem.
-
Strengthen relationships between immunization information systems (IIS), electronic health record systems (EHRs) health information technology (IT), health information exchanges (HIEs), clinicians and public health
-
Raise awareness of the issues that challenge immunization interoperability and create resources to reduce their impact
-
Develop recommended guidance and solutions for prioritized issues
-
Drive adoption of recommended solutions that benefit the immunization ecosystem
-
Current priorities
The IIP Collaborative, comprised of IIS, EHRs and other health IT, HIEs, clinicians, public health agencies, pharmacy and other critical partners, is addressing identified priorities for 2025 to improve immunization interoperability, information sharing and management including:
-
Immunization data from pharmacies
Pharmacies play an important role in immunization. We want pharmacy systems to be able to leverage immunization data exchange standards and best practices to improve interoperability with IIS and maximize successful immunization submission.
-
Submissions prior to the assignment of a legal baby name
The first vaccination in a person’s life, hepatitis B, typically happens within 24 hours of birth. Sometimes, the legal name has not yet been assigned, and when an HL7 immunization message is sent from the hospital to the IIS, the name is often just a placeholder (e.g., BabyBoySmith). When the legal name is available, the hepatitis B record needs to be identified and updated to avoid duplicate records.
-
Triggers that prompt submission or query messages from EHRs to IIS
IIS usability could be improved through optimally defined and timed submission and query triggers. If health IT systems can exchange data with IIS at appropriate times based on specific criteria, patient data will be available to providers when needed during clinic workflows and unnecessary queries to the IIS can be avoided.
-
Share your issue
The IIP is always accepting issues as trends change and new issues emerge in the immunization data exchange ecosystem.
-

-
Our approach to prioritization
The IIP solicits immunization interoperability challenges from the broader immunization ecosystem and works with CDC to screen the issues to ensure topics are within scope not duplicative of other efforts. The IIP Executive Council scores the issues based on criteria listed above to inform the IIP and assist the CDC Informatics and Data Analytics Branch (IDAB) to select issues to tackle.
The goal is to develop solutions to high-priority immunization interoperability issues that are feasible to implement and will have a positive impact on quality, safety, interoperability and information sharing.
IIP solutions
See how we address inefficiencies within healthcare to improve immunization integration.
-
Regularly updating code sets
Out of sync vaccine-related code set values (e.g., NDCs for annual flu vaccine or new vaccines) between immunization data exchange partners impedes data interoperability and can prevent patients’ vaccinations from being recorded properly. The Code Sets work effort launched in fall 2022 to collaborate with key partners to develop recommendations. This project seeks to develop resources to reduce:
- The level of effort and length of time required to implement new values within vaccine-related code sets used for interoperability between IIS and other health IT systems
- The variation in understanding of code set values (e.g. use of specific v. generic, NOS codes) and code set metadata (e.g., “code is active”) between IIS and applications used by their trading partners (e.g., EHRs)
This effort builds upon the work done in AIRA’s 2020 Vaccine Code Set Considerations. In the fall of 2023, information was gathered from 50,000+ individuals who currently use CDC provided vaccine code set resources in the form of an online poll — once the poll is live, a link will be provided here. Contact us at iip@himss.org to learn more about the topic, share your expertise or participate in solution development. -
Data validation and addressing reasons for IIS rejections
- Inconsistency in how data is entered at the point of registration or vaccine administration such as dates entered in the future, birth date discrepancies, or missing fields can cause IIS to reject immunization messages. Assessing data for validity, completeness and accuracy before submission facilitates successful submission and reduces the amount of rejected data that must be investigated, corrected, and resubmitted.
- The IIP Data Validation Workgroup developed comprehensive strategies to improve successful immunization submission to IIS at the point of care including end-user training resources and a common data error template for customization by IIS. Contact us at iip@himss.org to learn more and explore technical assistance opportunities.
-
Accessing bulk query data from IIS
- On average, IIS receives twice as many queries as submissions. Key partner groups are unable to access the data they need in a timely manner because of disparate and/or inadequate query or bulk data protocols.
- The IIP Collaborative Bulk Query Workgroup developed a bulk query toolkit which includes best practice guidance for bulk query options. Contact us at iip@himss.org to learn more and explore technical assistance opportunities.
-
Acknowledgments message dandling and error report guidance
The IIP Acknowledgments Workgroup developed a guidance document and implemented a pilot program to promote and validate the guidance. The workgroup convened to address variability in acknowledgment message handling among EHRs, IIS, and clinicians to improve the quality of information used for decision making.
Technical assistance opportunities are available to implement the Acknowledgments Guidance with interested partners. Email iip@himss.org to learn about technical assistance opportunities.
-
Multiple match query solution
The IIP Collaborative Multiple Match Query Workgroup developed a functionalities catalog and aggregate measures for multiple matches to improve the way in which multiple patient matches, from a query to an IIS, are handled among clinicians, clinical software systems, and HIEs.
-
Patient matching brief
- The IIP developed an educational brief to raise awareness around patient identification and matching to support immunization information sharing and management. When published, the educational brief will serve to inform:
- Current national patient matching strategies to emphasize the need for faster policy change
- Implications of the current unique health identifier federal funding ban
- Provide unbiased education as a community of experts providing insight and consensus around immunization
- Discussion supporting patient matching strategies
- The IIP developed an educational brief to raise awareness around patient identification and matching to support immunization information sharing and management. When published, the educational brief will serve to inform:
-
Common standard for transport of immunization messages
- The IIP Executive Council gained agreement on the unified adoption of a standard transport protocol to improve interoperability and information sharing among EHRs and IIS to support public health immunization goals.
-
Learn more about IIP solutions
The IIP offers technical assistance and support to immunization data exchange partners who would like to implement our recommendations. Contact us at iip@himss.org to learn more about how to engage with us to implement IIP solutions.
-
-
Meet the IIP executive council
The IIP is supported by an executive council of leaders that represent key collaborators.

-
We can improve health by advancing state-of-the-art immunization registries. I'm engaged with IIP because I'm motivated to positively impact population health.
Shaun Grannis, MD, MS
Vice President for Data & Analytics Regenstrief Institute, Inc.
-
The IIP work will continue to improve accurate, standards-based exchange of immunization information for use in patient care and public health.
Amit Popat
Integration Standards & Regulations Epic
-
The ability to interact and learn from others in this space is very useful. It's great to strengthen professional networks- as another benefit to the program.
Craig Newman
Interoperability Standards Analyst Altarum
-
Participation has allowed me to represent the pediatric clinical voice in important immunization information exchange conversations. We need your voice, too.
Susan Kressly, MD, FAAP
Pediatric Health IT SME
-
The IIP adds value to the entire industry and holds us accountable for improving immunization integration in the EHR, setting an example for HIT validation.
Kristen Glaza
Sr. Manager, Product Management Oracle Health
-

Scott Kirby
Physician’s Computer Company (PCC) Co-Chair
-

David McCormick
Indiana State Department of Health Co-Chair
-

Lisa Bari, MBA, MPH
Civitas Networks for Health
-

Mike Berry
HLN Consulting
-

Theresa Case, RN
athenahealth
-

Sarah Corley, MD
MITRE
-

Shannon Coleman, MS
STChealth
-

Cheryl Lee Eberting, MD
AZOVA
-

Kristin Glaza
Oracle Cerner
-

Geoff Gross
Oracle Cerner
-

Amanda (Mandy) Harris
Nevada Department of Health and Human Services
-

Steve Hill
Oracle Cerner
-

Baskar Krishnamoorthy
Department of Health, Immunization Section, Florida SHOTS
-

Susan J. Kressly, MD
Pediatric Health IT SME
-

Becky Learn
Indiana Health Information Exchange
-

Brian Lee
STChealth
-

Diane McLeod
Department of Health, Immunization Section, Florida SHOTS
-

Steve Murchie, MBA,
Envision
-

Craig Newman, PhD
Altarum
-

Michael Perretta
Docket ®
-

Amit Popat
Epic
-

Michael Powell, MSc
California Department of Public Health
-

Jon Reid, MBA
Utah Department of Health
-

Elizabeth Ruebush, MPH
Association of State and Territorial Health Officials
-

Pamela Schweitzer, RADM (ret.), PharmD
Pharmacy Leader and Expert
-

Nathan Scott
Docket®
-

Ron Shapiro
Qvera
-

Kimberly J. Stanislo, DNP, APRN, CPNP-PC, LSN
National Association of School Nurses (NASN)
-

Srinivas Tadikonda, MPA
Gainwell Technologies
-

Kristy Westfall
Association of Immunization Managers
Ex-officio members
Lynn Gibbs-Scharf, MPH, CDC
Kafayat Adeniyi, MPH, CDC
Stuart Myerburg, JD, CDC
Christine E. Miner, MPH, CDC
Katie Tully, MSPH, HHS/ONC
John Bender, MPH, MS, HHS/ONC
-
-
History of the Immunization Integration Program (IIP)
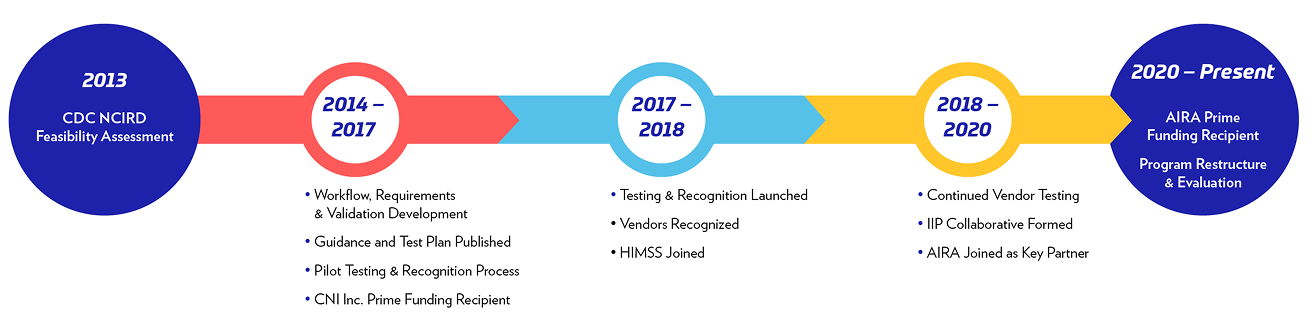
In 2013, following an extensive feasibility study conducted as part of its strategic planning process, the Informatics and Data Analytics Branch (IDAB) of the Center for Disease Control and Prevention (CDC) launched a project to improve immunization management and information sharing across electronic health records (EHRs) and immunization information systems (IIS) through a contract with Chickasaw Nation Industries (currently “Chickasaw Health Consulting, LLC,” previous contractor with the IIP).
HIMSS joined the project in early 2017 to launch the independent, voluntary IIP Testing and Recognition initiative — supporting the integration of immunization-related capabilities within EHRs and other clinical software to improve immunization management and information sharing. In 2019, HIMSS, AIRA, Drummond and Chickasaw Health Consulting launched the IIP Collaborative to identify challenges associated with EHR-IIS immunization information sharing and management and develop the adoption of solutions to address those challenges.
In 2020, the American Immunization Registry Association (AIRA) assumed leadership of the program and continues to partner closely with HIMSS, Drummond Group and additional experts in the immunization interoperability field. The IIP works closely with CDC and is part of a broader effort at CDC IDAB to use information technology to improve vaccination coverage and reduce vaccine-preventable disease.Frequently asked questions
Everything you need to know about the HIMSS Immunization Integration Program (IPP).
-
What is the Immunization Integration Program (IIP)?
The IIP, implemented by HIMSS, AIRA, and Drummond Group under a cooperative agreement with CDC IDAB, brings together IIS, EHRs and other health IT, HIEs, clinicians, public health, pharmacy systems, and other critical partners to improve immunization interoperability, information sharing and management.
-
What are the goals and key components of the IIP?
By improving immunization interoperability and functionality, the IIP aims to improve data quality spanning from the point of care to public health with the longer-term goals of increasing vaccination coverage and reducing vaccine-preventable diseases. The IIP achieves its goals in two primary ways:
- The Testing and Recognition initiative, which facilitates the adoption of consensus immunization-related capabilities within EHRs and other clinical software to improve clinical decision-making and information sharing with IIS.
- The Collaborative which brings IIS, EHRs and other health IT, HIEs, clinicians, public health and other critical partners together to develop and drive the adoption of solutions to improve interoperability, information sharing, and access to immunization information by clinicians and public health.
-
What is the purpose of the Collaborative?
The Collaborative plays a key role in the project’s efforts to improve information sharing and enable access to immunization information among IIS, EHRs and other health IT, HIEs, clinicians, public health and other critical partners.
The purpose of the Collaborative is to identify challenges associated with EHR-IIS immunization information sharing and management and to develop and drive the adoption of solutions that address those challenges.
-
What is the Collaborative’s approach?
The IIP uses the following approach to accomplish its goals:
- Strengthen relationships among IIS, EHRs and other health IT, HIEs, clinicians, public health and other critical partners via a governance structure that includes these essential perspectives.
- Identify issues associated with interoperability and information sharing and management.
- Develop recommended solutions of the prioritized issues
- Create resources to raise awareness of the impact to the issues challenging immunization interoperability.
- Leverage channels to drive adoption of recommended solutions and their benefits to the immunization ecosystem
-
What is the purpose of the Testing and Recognition Initiative?
The program facilitates adoption of consensus immunization-related capabilities within EHRs that improve clinical decision-making, reduce burden and improve information sharing among vaccination providers and IIS by:
- Facilitating agreement on priority immunization-related software capabilities that improve clinical decision-making, reduce burden and improve information sharing among IIS, EHRs and other health IT, HIEs, clinicians, public health and other critical partners.
- Operating a voluntary testing and recognition initiative that enables EHR and other software developers to both integrate and demonstrate their products’ inclusion of immunization-related capabilities
-
What does IIP software recognition mean?
Products that have received IIP recognition have successfully tested against immunization-related software capabilities across eight clinical domains:
- Registering and identifying a patient (to assure the provider and the registry are communicating about the same patient).
- Managing external query, response and reconciliation (to enable bidirectional data sharing with immunization registries, providing more accurate data for managing patient care).
- Managing information for clinical decision-making (to enable the right immunization at the right time for the right patients).
- Managing inventory (to assist with managing private vaccine and government guarantee program stock).
- Administering and reporting immunizations (to assure accurate, non-obtrusive data entry, enabling patient management and reporting to immunization registries).
- Managing cohorts of patients (to encourage timely vaccinations and help manage any vaccine recalls).
- Managing adverse events (to identify safety issues, help prevent future adverse events, and support reporting).
- Providing patient access to immunization information (to help patients and their caregivers access their immunization histories)
-
How does the IIP Testing & Recognition initiative relate to the (ONC) Health IT (f)(1) Transmission to immunization registries Certification Program?
- The IIP has been approved by the ONC as an alternative testing method for the ONC Health IT Certification Program.
- Five of the core testing requirements under the IIP align with Certification Criteria within (f)(1) Transmission to Immunization Registries within the ONC Health IT Certification Program.
- IIP’s approval by ONC as an alternative testing method means that those software products that achieve IIP recognition can “get credit” for having successfully tested against the ONC Health IT Certification (f)(1) Certification Criteria.
- ONC-Approved Health IT Certification bodies can now rely upon IIP Recognition, and therefore, will not need to also test for compliance with ONC’s (f)(1) Certification Criteria.
-




















